A week of LaTeX related activities
Last weekend July 24-26, 2020, saw this years TUG conference go virtual. This was great for those of us who can't normally attend these sorts of events.
A good few interesting and useful talks. I didn't attend all of them, but the ones I did included:
- Eric Mc Sween – Integrating a new TeXLive in to Overleaf
- Jim Hefferon – A new set of LaTeX packages
As the videos are going to be put online, I will catch up lter. Joseph Wright's talk on LearnLaTeX is interesting too.
Following this, on Thrusday 30th July I attended the webinar on “How to Create Professional Looking Documents In-House”. This was interesting but a little bit beyond my use case.
However it did demonstrate what Overleaf is capable of, so something I would recommend to others.
The webinar also refereed to the Jupyter editor which again looks interesting. The Overleaf webinar, will be available online soon.
In all an interesting week, I need to catch up on the talks, I have downloaded the pdf on LaTeX packages, as I took notes so now have a nice list of useful packages that can serve quite a few use cases when creating documents.
#latex
Mars Perseverance Rover
The Mars Perseverance Rover launched today in the Atlas V rocket, as planned. You can read more about this on the NASA mission page.
Should arrive on Mars in early 2021
Feel free to discuss further on the QOTO Discourse forum.*
- Free sign up will be required.
#science,#space,#Mars,#rover,#mission,#NASA,
Overleaf Webinar 30th July 2020
Todays Overleaf webinar covers
How to Create Professional Looking Documents In-House
Date : Thursday 30 July
Times (UTC): 11:00am ET/3:00pm UTC/08:00am PT
Sign up for free here
#Overleaf, #webinar, #files, #professional, #document, #creation
New Citizen science project from Zooniverse
I was sent this on 21st July 2020
We're happy to announce the launch of a new project:
* A Century of Natural History Observations
For an explanation of the project, let's hear from the project team:
“For over a century, the team at Mohonk Preserve has been diligently collecting data on all aspects of the natural life in the Shawangunk Mountains region.
Everything from weather patterns to the phenology of local species is
carefully monitored and catalogued each year, creating a treasure trove of data for anyone who wants to know more about the phenology of the “Gunks.” Today we use technology to store and track our data, but this was not always the case. Daniel Smiley, the creator of this project, originally would walk the Shawangunk Ridge lands with notecards in hand, writing down whatever he saw. Over the course of his lifetime, he and other researchers with whom he worked created over 10,000 notecards.
These notecards are rich with detailed information that is useful in a
multitude of research projects in areas like ecology, land-use change,
animal migration, and species’ range changes. Rather than keeping this
important research tool tucked away in file cabinets, we want to make these cards digitally available to scientists, students and all who want to use them. Through a generous grant from the Institute of Museum and Library Services through the Museums for America program, we’ve been able to establish a project protocol and create digital images of over 9,000 Dan’s note cards. Now we’re asking you for help making this information fully searchable by looking at individual cards and transcribing the information on them. Thank you for helping us share and expand access to this remarkable natural history resource!”
Learn more and get involved at:
https://www.zooniverse.org/projects/mohonkpreservedsrc/a-century-of-natural-history-observations
Thanks for all your help!
Cliff & the Zooniverse Team
Chances
Jonaed Iqbal
Jonaed Iqbal
The Harvard debate team went against a prison debate team.
Who do you think won?
The Prison debate team!
Isn't that crazy?
Not only did they beat Harvard, they also beat the University of Vermont, West point and the University of Cambridge.
These are prepared debates where both sides are given a position that they have to defend.
The university students have access to the internet.
The prisoners only have access to books in their prison's library.
Some of them don't even have their GED.
They are competing with students that typically grew up in wealthy households.
Some of the people they are competing with have started debating at 4 years old.
It just goes to show that you really shouldn't count anyone out because of their background and their lack of formal education.
It's a beautiful site watching debaters in state issued green jump suits wearing bow ties borrowed from their fellow inmates beat opponents dressed in black suits.
Jonaed
NoDegree
#personaldevelopment #motivation #education #inspiration #nodegree
More molecular modelling
So following on from my Post on 23/7/2020 I decided to share some of the info graphics created by Compound Chemistry
These are useful for GCSE and A'level chemistry. Building the models could help enforce the learning provided. I would strongly suggest talking to your teacher for advice on this.
So first off, looking at how different organic molecules are named.
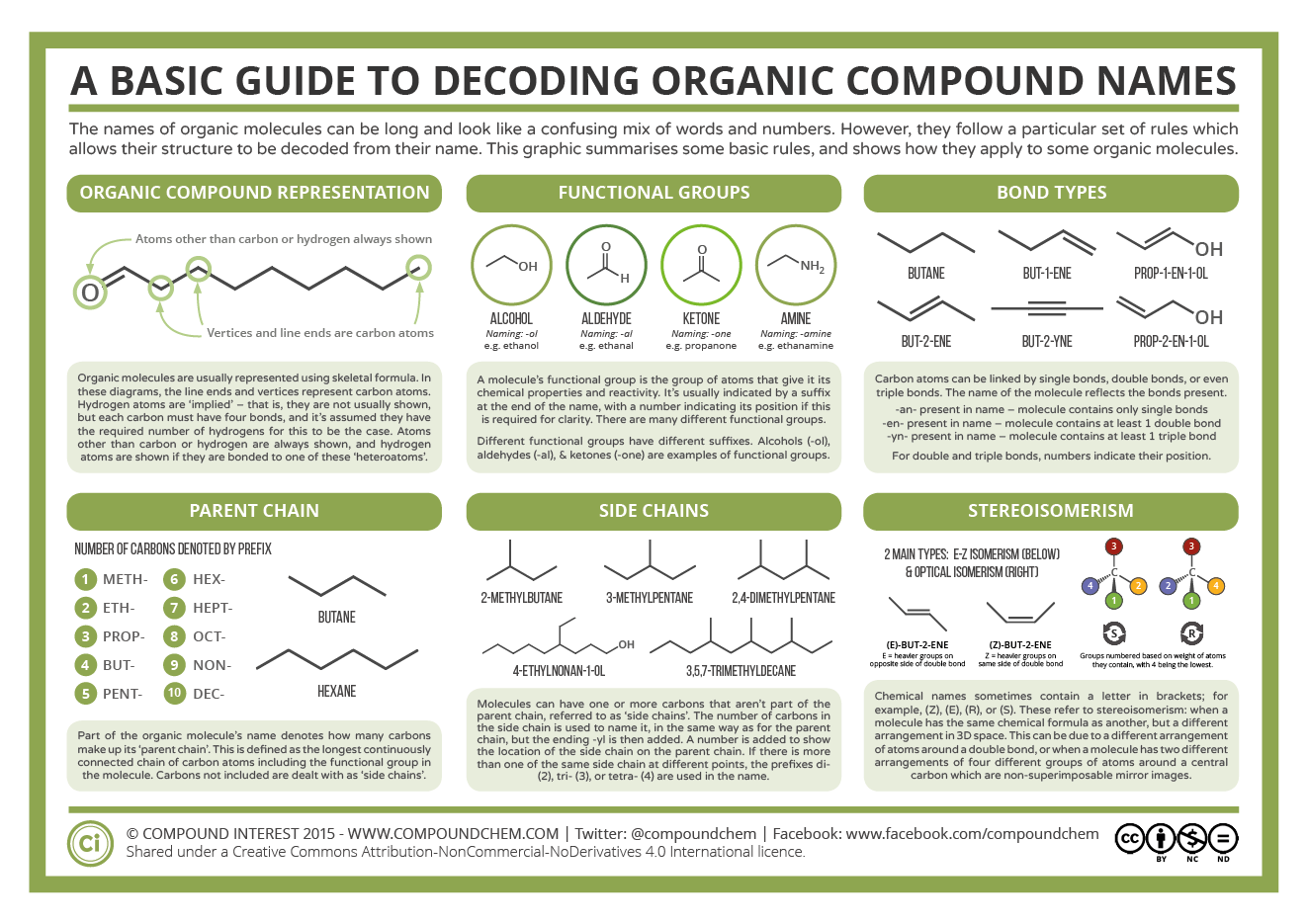
So you should be able to take some of the diagrams here and create some basic models. Don't forget to you can use the bendy straws for double bonds.
As isomerism is also covered in the curriculum (or was) this may also be useful.

There are lots of infographics on the website listed below.
#chemistry,#organic,#compoundchemistry,#modeling,#clay,#education.
Molecular weight calculator
I decided to write this to help calculate molar weights for chemistry.
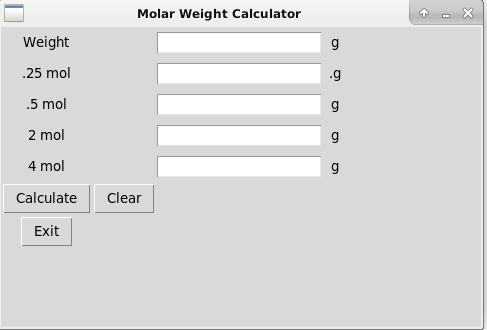
In essence you can enter the Mass of an element or molecular mass of a substance, compound etc, and this will help give you molar weight, for example 0.5 mol.
It is, for example common to have different concentrations of an acid for example. We know that Sodium Hydroxide has a weight of 40 because of the combined weight of its components:
$NaOH$ which equates to
Na = 22 +
O = 16 +
H = 1
= 39
Therefore 1 mol of NaOH = 39g which is of course equal to Avogadros constant : $6.022 x 10^{23}$
Therefore 0.5 mol is roughly $39 \div 2 = 19.5g$
This program is NOT a substitute for proper calculation. You need to use more accurate values. Values used are just a rough guide.
However it may be useful, for those quick calculations.
The program code base is taken from my Drake equation calculator I made a few weeks ago.
#chemistry,#mol,#molar,#weight,#calculator
Molecular modelling with clay part 3
Further to the post earlier I am having mixed results trying to make double bonds from bendy straws. This is really proving to be not the most ideal solution.
I am experimenting with using straight straws side by side to represent a double bond.
This sort of works, but still isn't ideal, perhaps I'll just stick to single bond molecules.
Doing modelling with clay is certainly not an ideal solution but, as stated it may simply help with some basic modelling if you can't get hold of the actual modelling kits.
Just don't try and be adventurous.
This is science, so it needs some more experimentation to find an ideal solution.
if anyone has those craft match sticks such as these:-

as sold by Factory Direct Craft.com
This may prove to be a better solution than straws. You can get shorter lengths which will probably be better.
Don't go and buy any, I would suggest to see if you can get a few to experiment with, after school or other child care clubs usually have craft materials, so I would suggest experimenting first.
#chemistry,#chemical,#modelling,#clay,#straws,#visualisation
Molecular modelling with clay part 2
As discussed in my post yesterday. I am going to introduce how to build a few a basic molecules with the resources suggested.
Step 1
Firstly I have cut the Black and White modelling clay to size.

The sections on the RIGHT can be put away for later use. You can see I have more White clay for Hydrogen than Black which I am using for Carbon.
Step 2
Now cut the clay up, roll in to balls and assemble the straws (links) that are needed (in this case 4) It is a good idea to make sure you have all the bits you need to hand.

The Carbon is bigger as we need to attach the 4 Hydrogen to this.

So here, is our Methane model, partly assembled. I have left the last one off for illustration.

The final step is to attach the last Hydrogen to the Carbon creating Methane $CH_4$.
Hope this helps, it may not be perfect but it may help if you can't get hold of actual Molymod kits.
Hopefully what this will allow you to do is visualise basic chemical structures.
There is an addition to this post here
#chemistry,#chemical,#modelling,#clay,#straws,#visualisation
Hubble – Eye in the Sky part 3
Part 3 of the NASA Mission Operations – Hubble Video Miniseries is released today.
The series so far, has been really interesting and each episode is roughly 10 minutes in length, so could make an ideal teaching resource too.
#nasa,#hubble,#space,#telescope,#miniseries,#documentary,#EyeintheSky



